Anti-Inflammatory Protein Therapy After Heart Attack: A Breakthrough Strategy to Reduce Cardiac Damage and Heart Failure Risk
Introduction: Why Inflammation After a Heart Attack Still Matters
Myocardial infarction (MI), commonly known as a heart attack, remains one of the leading causes of morbidity and mortality worldwide. Despite major advances in acute interventions—such as rapid coronary reperfusion using percutaneous coronary intervention (PCI)—many patients still progress to chronic heart failure.
A critical contributor to this progression is excessive post-infarction inflammation. While inflammation is essential for clearing dead tissue and initiating repair, an uncontrolled inflammatory response can lead to:
- Additional loss of viable heart muscle
- Excessive fibrotic scarring
- Reduced long-term cardiac function
This has driven growing interest in targeted anti-inflammatory therapies that could preserve heart tissue without impairing healing.
The Role of Inflammation in Myocardial Infarction
Following coronary artery occlusion, oxygen deprivation causes ischemic injury to cardiomyocytes. Restoration of blood flow—although lifesaving—paradoxically triggers ischemia-reperfusion injury, amplifying inflammation.
Key Inflammatory Processes After MI
- Activation of neutrophils and macrophages
- Release of pro-inflammatory cytokines and chemokines
- Increased vascular permeability
- Infiltration of immune cells into injured myocardium
While necessary for debris clearance, this response often results in secondary myocardial damage, accelerating ventricular remodeling and heart failure development.
Anti-Inflammatory Proteins: A Novel Therapeutic Direction
A recent study published in The American Journal of Pathology provides compelling evidence that anti-inflammatory protein therapy may offer a new way to limit post-MI damage.
Researchers investigated two biologically active molecules:
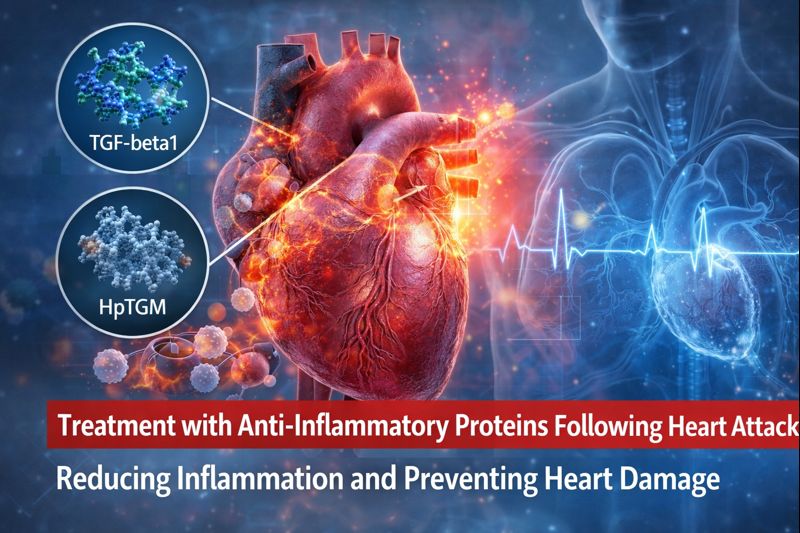
- Transforming Growth Factor-β1 (TGFβ1) – an endogenous protein released following tissue injury
- Heligmosomoides polygyrus TGM (HpTGM) – a parasite-derived protein that mimics TGFβ signaling to suppress host inflammation
Key Findings
- Both TGFβ1 and HpTGM significantly reduced inflammatory cell infiltration in injured heart tissue
- Treatment resulted in smaller mature scar size
- Long-term structural damage to the heart was reduced
These results suggest that modulating—not eliminating—post-infarction inflammation may be the key to improving outcomes.
Mechanism of Action: How These Proteins Protect the Heart
Despite their very different biological origins, TGFβ1 and HpTGM demonstrated nearly identical therapeutic effects.
Shared Mechanistic Pathway
- Activation of the TGFβ signaling pathway
- Suppression of excessive immune activation
- Protection of endothelial cells, which regulate immune cell entry into injured tissue
By stabilizing the vascular endothelium, these proteins limit the uncontrolled migration of pro-inflammatory white blood cells, preventing unnecessary collateral damage.
Importantly, therapy was administered at the time of reperfusion, aligning well with real-world clinical workflows for heart attack treatment.
Why HpTGM Is Especially Exciting for Drug Development
While TGFβ1 has long been recognized for its anti-inflammatory properties, its pleiotropic biological effects raise safety concerns in chronic use.
HpTGM, by contrast, is a parasitomimetic biologic evolved specifically to suppress inflammation without triggering broad systemic effects. This makes it a highly attractive candidate for translational and clinical development, particularly as a short-term therapy following acute MI.
Implications for Clinical Development and Regulatory Strategy
From a clinical development and regulatory affairs perspective, this research represents a meaningful shift:
- Focus on post-reperfusion intervention
- Use of biologic anti-inflammatory agents
- Targeting endothelial-immune interactions rather than cardiomyocytes alone
These findings may support:
- First-in-human translational studies
- Development of biologic or protein-based cardiovascular therapies
- Regulatory pathways emphasizing unmet medical need and disease modification
Conclusion: A Promising Step Toward Preventing Post-MI Heart Failure
This study provides strong preclinical evidence that anti-inflammatory protein therapy could become a transformative strategy in the management of myocardial infarction.
By selectively dampening harmful inflammation—without blocking essential healing processes—proteins such as HpTGM may help preserve heart function, reduce scarring, and lower the long-term risk of heart failure.
Further translational and clinical studies will be critical, but the therapeutic concept represents a significant advancement in cardiovascular drug development.
References
- Redgrave RE, et al. (2023). Exogenous Transforming Growth Factor-β1 and Its Mimic Heligmosomoides polygyrus TGM Attenuate the Heart’s Inflammatory Response to Ischemic Injury and Reduce Mature Scar Size. The American Journal of Pathology. Available at: https://ajp.amjpathol.org/article/S0002-9440(23)00376-0/fulltext.
- News-Medical (2024). Anti-inflammatory therapy shows promise following acute myocardial infarction. Available at: https://www.news-medical.net/news/20240404/Anti-inflammatory-therapy-shows-promise-to-treat-patients-following-acute-myocardial-infarction.aspx.
- Huang S, Frangogiannis NG. (2018). Anti-inflammatory therapies in myocardial infarction: failures, hopes and challenges. British Journal of Pharmacology, 175(9), 1377–1400. Available at: https://doi.org/10.1111/bph.14155.
Johns Hopkins Medicine (2022). Fight Inflammation to Help Prevent Heart Disease. Available at: https://www.hopkinsmedicine.org/health/wellness-and-prevention/fight-inflammation-to-help-prevent-heart-disease.