From Data to Submission: Turning Clinical & Safety Information into Regulatory Success
Bringing a therapy from development to approval is not only a scientific journey, it is a documentation journey. Health authorities do not approve intentions, effort, or potential. They approve clear, consistent, traceable evidence.
In 2026, agencies such as the FDA, EMA, and Health Canada expect submissions to demonstrate seamless alignment between protocol design, statistical planning, study conduct, analysis, and clinical interpretation. Gaps, contradictions, or unclear narratives can delay approvals, trigger major questions, and erode reviewer confidence. Regulatory success depends on how effectively clinical and safety data are transformed into a persuasive, defensible submission.
Why the Data Story Matters
Reviewers are not just checking numbers.
They are asking:
• Was the study designed appropriately?
• Were endpoints predefined and respected?
• Are analyses consistent with the SAP?
• Do results support the claims?
• Is safety adequately characterized?
If the answers are difficult to locate or inconsistent across documents, concerns escalate quickly. A successful submission tells one coherent story, repeated accurately across modules.
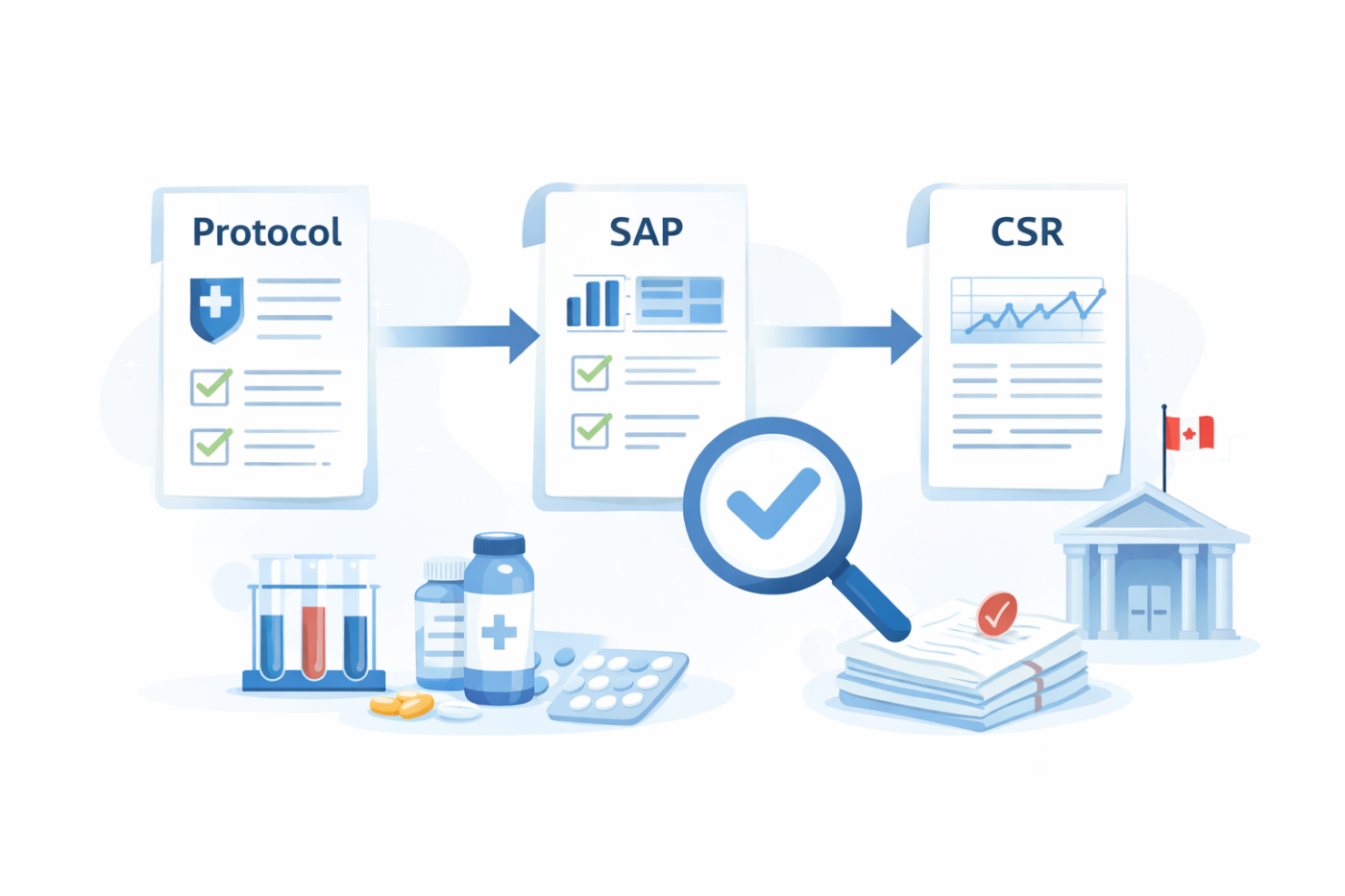
The Critical Data Flow: Protocol → SAP → CSR
Every approval begins with disciplined traceability.
Protocol
Defines objectives, endpoints, populations, and methodology.
Statistical Analysis Plan (SAP)
Specifies exactly how those endpoints will be analyzed.
Clinical Study Report (CSR)
Presents results and interprets them according to the SAP.
Breakdowns occur when:
• Analyses deviate without explanation
• Populations change between documents
• Endpoints are reinterpreted
• Post-hoc analyses dominate conclusions
Regulators expect transparency. Unexplained evolution looks like bias.
Consistency Across Modules: A Hidden Risk Area
Many teams underestimate how often reviewers cross-reference documents.
Examples:
• Efficacy statements in summaries must match CSR tables
• Safety language must align between narratives and listings
• Benefit-risk arguments must be reproducible from data
• Manufacturing or labeling claims must reflect clinical evidence
Even small mismatches create doubt. When confidence decreases, questions increase.
What Reviewers Expect in 2026
Health authorities increasingly work under accelerated timelines. They depend on sponsors to provide clarity.
Reviewers value:
• Logical structure
• Rapid navigation
• Clear definitions
• Transparent derivations
• Explanation of deviations
• Well-supported conclusions
They should never have to “guess” what the sponsor means. A well-prepared submission reduces reviewer workload and that builds trust.
The Most Common Submission Deficiencies
Across agencies, similar issues appear repeatedly.
Traceability Gaps
Inability to connect objectives → analyses → conclusions.
Inconsistent Numbers
Different values for the same endpoint in different sections.
Overinterpretation
Claims that exceed statistical support.
Weak Safety Integration
Failure to present a comprehensive, contextualized safety profile.
Late-Stage Document Assembly
Trying to harmonize materials just before submission.
These problems are preventable with early planning and cross-functional alignment.
Medical Writing Is Strategic, Not Editorial
High-quality medical writing is not formatting.
It requires:
• Understanding regulatory logic
• Anticipating reviewer questions
• Protecting scientific credibility
• Ensuring internal consistency
• Presenting complex data clearly
Writers must function as translators between science, statistics, medicine, and regulation. When done correctly, they dramatically reduce review cycles.
How Strong Preparation Accelerates Approvals
Organizations that succeed share common practices:
• Integrated document planning early in development
• Templates aligned with regulatory expectations
• Structured review workflows
• Predefined data verification
• Cross-team communication between clinical, biostatistics, safety, and regulatory
They build submission readiness years, not months, before filing.
Conclusion
Regulatory agencies expect precision, clarity, and accountability. When clinical and safety information is integrated effectively, submissions become stronger, questions decrease, and approvals move faster. Regulatory success is built on disciplined storytelling backed by data.